Engineering the PFAS-Free Future: UHMWPE as a High-Performance Alternative to PTFE Liners

For years, PTFE has been the default choice for catheter liners. It’s well understood, widely used, and delivers the lubricity R&D engineers rely on.
But PFAS scrutiny is intensifying worldwide, putting pressure on PTFE supply chains and pushing the MedTech industry to evaluate alternatives.
For R&D teams, that raises practical questions. What does this mean for your designs, and what viable alternatives should you be evaluating now?
In this webinar, Cheng Wei, Sr. Scientist and Application Expert at dsm-firmenich, joins Katie Karmelek, Chamfr Co-Founder, to break down these questions, focusing on UHMWPE as a high-performance alternative to PTFE liners.
Want to watch the full session? Sign up to access the webinar recording →
Why PFAS Scrutiny Is Shaping Material Decisions
One of the challenges highlighted in the discussion is how difficult it is to interpret what PFAS actually means for device development today.
There are a few key factors driving increased attention, making the landscape harder to navigate:
- Regulatory activity in Europe evaluating broader PFAS restrictions
- Ongoing discussion across agencies, including FDA and EPA
- Shifts in supply as industries outside MedTech move away from PFAS
As mentioned in the webinar, the key question is not whether PTFE should be replaced immediately. It’s how teams should think about material decisions as requirements, supply, and expectations continue to evolve.
A New Option: UHMWPE as a PFAS-Free Alternative
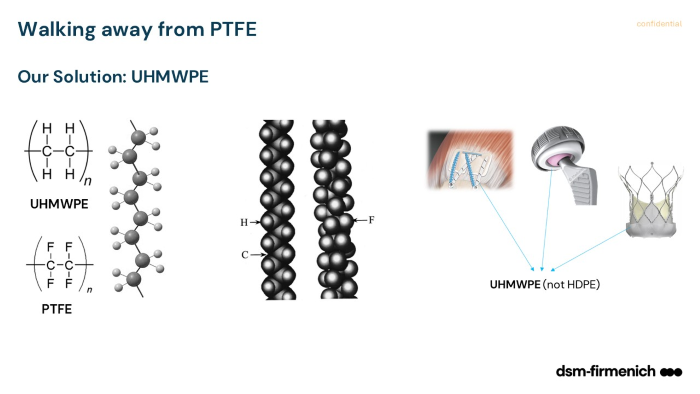
To address these pressures, the webinar introduces ultra-high molecular weight polyethylene (UHMWPE) as a potential alternative to PTFE liners.
What makes UHMWPE interesting is how closely it aligns with what engineers already need, while introducing a different set of trade-offs.
From a material standpoint, it offers:
- Low coefficient of friction
- Strong biocompatibility and chemical resistance
- Established clinical history in other applications
It’s important for engineers to understand how it behaves within their specific design, especially across processing conditions, bonding, and final device performance.
The webinar also highlights where UHMWPE liners may be a strong fit, including introducers, neurovascular catheters, and steerable devices.
Where Material Trade-Offs Become Critical
One of the most valuable aspects of the discussion was how UHMWPE reframes material selection beyond a single property.
PTFE is often chosen for its lubricity. But in practice, device performance depends on a broader set of factors. For early-stage teams, the discussion highlights how material differences show up beyond just friction.
Key considerations include:
- Scratch resistance, which impacts durability and surface integrity
- Bond strength to the outer jacket, affecting device construction
- Processing behavior, especially during reflow and manufacturing
For example, UHMWPE demonstrates significantly higher scratch resistance compared to PTFE, as shown in the webinar data.
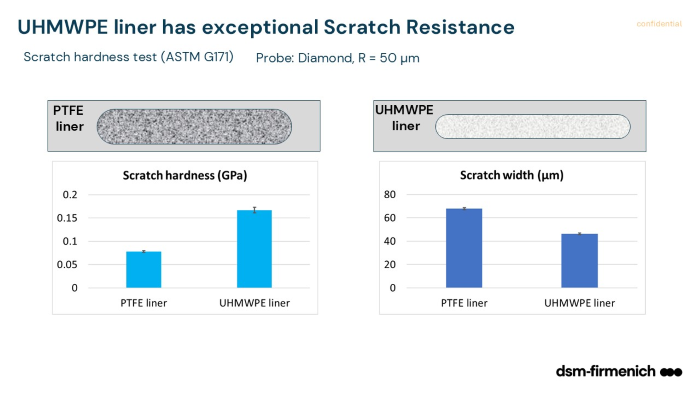
These differences influence how the material performs within the full device, not just at the material level.
Manufacturing Implications That Impact Design
A key difference highlighted in the webinar is how UHMWPE behaves during processing.
Unlike PTFE, which remains stable during reflow, UHMWPE softens and flows. This changes how engineers need to think about:
- Final dimensions after reflow
- Surface morphology and roughness
- Process control and repeatability
It also introduces alternative manufacturing approaches. As shown in the presentation, UHMWPE liners can be produced using non-extrusion methods, enabling:
- Thin wall capability, typically in the 20–50 micron range
- Flexibility across a wide diameter range
- Different stability characteristics during processing
For teams used to PTFE-based workflows, these differences require early consideration.
Material Selection Doesn’t Stop at Performance
Material selection extends beyond core properties. Two key areas stood out in the discussion:
Sterilization and Durability
UHMWPE demonstrates promising compatibility with common sterilization methods, including gamma and EtO, without significant degradation in mechanical performance under certain conditions.
Sustainability and Long-Term Strategy
Sustainability is increasingly moving from a reporting metric to a design consideration.
Teams are already seeing:
- Procurement requirements incorporating sustainability criteria
- Increased attention on emissions and material sourcing
- Broader evaluation of long-term environmental impact
For engineers, this adds another layer to material selection decisions, especially in early development.
Final Thoughts on Rethinking PTFE in Early Development

PTFE remains a critical material in many devices today. The pressure around PFAS is not because PTFE no longer works, but because future requirements around supply, manufacturing, and material selection continue to evolve.
For R&D teams, that makes early evaluation more important.
Understanding how materials like UHMWPE behave in processing, bonding, and final device construction can help teams make more informed decisions before they are forced into them later.
This webinar offers a deeper look at those considerations and how teams can start evaluating alternatives earlier.