Reimagine Next-Gen Catheter Design with EverGlide+: A PFAS-Free, Sterilization-Stable Liner Platform
In this sponsored technical blog post, Dynaflex Technologies introduces EverGlide+, a PFAS-free, sterilization-stable liner platform engineered to help catheter R&D teams overcome bonding challenges, reduce delamination risk, and improve structural performance in next-generation shaft designs.
PTFE has been a foundational material in catheter design and development for decades. Its lubricity, thermal stability, and thin-wall capability have enabled generations of high-performance access, guide, and delivery systems. However, as catheter architectures evolve and global PFAS considerations come to the forefront, PTFE’s structural and regulatory constraints have become more significant, particularly in bonding-critical, high-torque, multi-durometer shaft constructions.
As catheter systems evolve toward thinner walls, higher torque transmission, and more complex multi-durometer architectures, liner materials are no longer selected based on lubricity alone. R&D engineers must increasingly balance lubricity with structural integration, sterilization compatibility, and long-term system reliability—especially as laminate integrity and distal durability become central to performance.
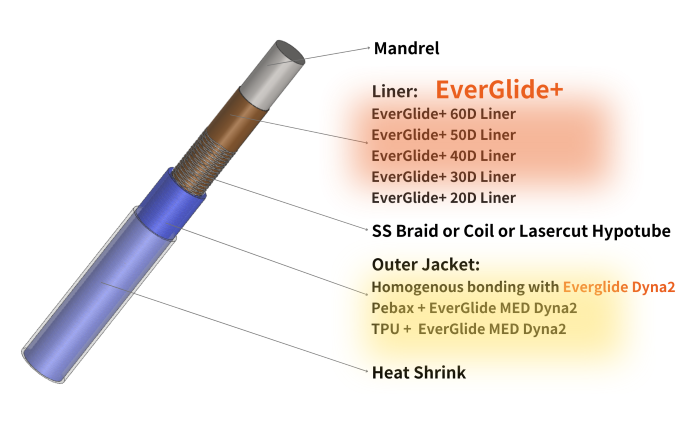
EverGlide+ is a PFAS-free, nylon-based liner platform designed to address these emerging challenges. It offers catheter-grade lubricity with stronger laminate bonding, improved torque transmission, and full compatibility with modern sterilization workflows. With durometers ranging from 20D to 60D, EverGlide+ supports a broad span of designs from soft distal microcatheters to high-pushability access systems.
Why PTFE Is Facing Pressure in Modern Catheter Design
PFAS Considerations
PTFE falls under PFAS classifications, and global efforts to reduce PFAS exposure have led many OEMs to evaluate long-term alternatives. While PTFE remains available today, forward-looking programs increasingly factor PFAS strategy into material selection.
Sterilization Limitations
PTFE cannot withstand E-beam sterilization and is generally restricted to EO. As more programs transition to E-beam for logistics and throughput benefits, PTFE becomes a limiting factor.
EverGlide+ is compatible with E-beam, EO, and hydrogen peroxide plasma (H₂O₂), enabling sterilization flexibility without redesigning around material constraints.
Slip-Layer Delamination
PTFE does not bond to braid/coil or jacket polymers, such as Pebax®, TPU, or nylon. This creates a slip layer within the laminate that may contribute to:
- Torque loss
- Delamination in LCHT zones
- Fatigue-related failures
- Reduced transition durability
These concerns grow as engineers adopt thinner walls and more complex multi-durometer structures.
The Enduring Strengths of PTFE
PTFE remains the most lubricious solid polymer, offers exceptional thermal stability, and supports the thinnest liner walls achievable in medical extrusion. These properties make PTFE indispensable in many current catheter systems.
EverGlide+ is not intended to universally replace PTFE. Instead, it provides a PFAS-free, structurally bondable alternative for applications where laminate integrity, sterilization flexibility, or distal-tip durability are core design considerations.
EverGlide+: A PFAS-Free Liner Platform Built for Structural Integration
EverGlide+ was engineered to deliver lubricity comparable to PTFE within a nylon-based material that bonds cohesively to braid and jacket layers during reflow.
Key Material Attributes
- PFAS-free
- Lubricity comparable to modified PTFE blends
- Bondable to braid and jackets (nylon-based)
- Available in 20D–60D
- High elongation and toughness
- Stable under E-beam, EO, and H₂O₂
- Smooth thermal transitions during reflow
Tunable Stiffness Across 2F–24F Devices
The durometer range enables intentional stiffness control:
- 20–30D: Atraumatic distal liners and LCHT zones
- 40–50D: General liner applications
- 60D: High-pushability proximal regions
Bonding through the laminate supports smoother stiffness transitions compared to PTFE-based constructions.
By integrating stiffness transitions into a bonded laminate rather than localizing them at slip interfaces, EverGlide+ helps distribute mechanical stress more evenly along the shaft. This results in more predictable bending behavior and improved durability in multi-durometer catheter designs.
Non-Delaminating Distal and LCHT Designs
EverGlide+ allows fully bonded transitions in soft distal segments without the slip-layer separation seen in PTFE constructions. This supports longer fatigue life and more predictable bending behavior in neurovascular and peripheral platforms.
Mechanical Behavior in Composite Shafts
Unified Laminate Structure
Because EverGlide+ bonds to braid and jacket materials during reflow, it forms a unified structure without a slip interface. This contributes to:
- Improved torque response
- Enhanced pushability
- Better bending recovery
- Higher fatigue resistance
- More durable transitions
In traditional PTFE constructions, the slip interface can introduce lag between applied torque and distal response, especially in thin-wall or high braid-density designs.
A unified laminate reduces this slip behavior, helping torque and push inputs transmit more directly along the shaft for more consistent handling.
Wall Thickness and Structural Performance
PTFE provides unmatched ultra-thin wall capability, typically 0.0004″–0.00075″. EverGlide+ currently supports ~0.001″ with continued development toward thinner profiles.
While PTFE can achieve thinner liner walls, overall catheter performance is governed by the total system wall. Engineers commonly maintain identical OD by redistributing wall thickness between liner and jacket.
For example, for a shaft with ~0.004″ total wall:
- PTFE: 0.0005″ liner + 0.0030″ jacket
- EverGlide+: 0.0010″ liner + 0.0025″ jacket
This retains the same final dimension while leveraging EverGlide+’s structural contribution.
As a bondable nylon-based thermoplastic, a thicker EverGlide+ liner can:
- Eliminate slip-layer behavior
- Enhance torque and push transmission
- Improve fatigue durability
- Strengthen distal transitions
- Reduce risk of braid exposure through the ID
These laminate-level benefits often offset PTFE’s thin-wall advantage in many shaft constructions.
In practice, final device performance is rarely determined by liner thickness alone. System-level wall redistribution preserves OD while enabling laminate integrity gains, often improving torque response and transition durability without changing the overall size envelope.
Sterilization Compatibility
Supporting legacy and next-generation sterilization approaches, EverGlide+ is stable under:
- E-beam
- EO
- H₂O₂ plasma
Applications Across 2F–24F
- Microcatheters (2–5F): Soft, lubricious, atraumatic distal liners
- Guide catheters (6–8F): Improved torque response and laminate integrity
- Large-bore access systems (10–24F): Strengthened composite structures
- LCHT distal sections: Bonded transitions
- Dilators & introducers: Predictable mechanical response
EverGlide-Dyna: Lubricity Enhancement for Jacket Polymers
EverGlide-Dyna is a lubricity-enhancing additive compatible with Pebax®, TPU, and nylon blends. It provides:
- Lower COF than base polymers
- Improved wet lubricity
- Non-leaching, durable performance
- No cracking or particle generation
EverGlide-Dyna and EverGlide+ share compatible chemistries and support cohesive system-level design.
A PFAS-Free, Structurally Integrated Catheter Materials Strategy
Together, EverGlide+ and EverGlide-Dyna provide OEMs with:
- PFAS-free materials strategy
- Stronger laminate bonding
- Improved torque and bending response
- Alternatives to coating-dependent lubricity
- Broader sterilization compatibility
- More consistent, simplified manufacturing pathways
For next-generation catheter platforms, material selection is increasingly a system-level decision—balancing performance, manufacturability, sterilization workflows, and long-term regulatory strategy rather than optimizing a single property in isolation.
Key Takeaways for Catheter R&D Engineers
PTFE remains essential in applications where ultra-thin walls and maximum lubricity dominate. EverGlide+ is not presented as a universal replacement. Rather, it offers a structurally integrated, PFAS-free alternative designed for catheter programs where laminate integrity, sterilization flexibility, distal durability, or PFAS strategy shape material selection.
In combination with EverGlide-Dyna, Dynaflex provides a cohesive materials ecosystem for designing next-generation catheter platforms with improved system-level performance and manufacturing reliability.
Explore the lineup on Chamfr, with nearly 200 SKUs available to ship in 1 to 2 business days.
FAQs
EverGlide+ is a PFAS-free, nylon-based liner platform engineered to bond directly with braid/coil and jacket polymers during reflow, improving torque, transition durability, and overall laminate integrity in catheter shafts.
No. PTFE still excels in ultralow COF and the thinnest possible liner walls. EverGlide+ is a variable durometer and bondable alternative for applications where structural cohesion, sterilization flexibility, or PFAS considerations matter.
EverGlide+ is compatible with E-beam, EO (ethylene oxide), and vaporized hydrogen peroxide sterilization, offering more flexibility than fluoropolymer-based liners.
EverGlide+ currently supports thin-wall extrusion around ~0.001″, with development work underway toward thinner profiles. Final catheter dimensions can be preserved by adjusting jacket wall thickness while benefiting from improved laminate bonding.
EverGlide+ can be produced in 20D, 30D, 40D, 50D, and 60D, enabling tailored performance for navigation, distal transitions, and high-torque sections across 2F–24F catheter systems.
EverGlide-Dyna is a non-leaching lubricity additive that can be blended into Pebax®, TPU, or nylon jackets to enhance internal or external lubricity where coating durability, outsourcing logistics, or particle testing are concerns.
About Dynaflex Technologies
Dynaflex Technologies is a trusted medical device contract manufacturer specializing in catheter solutions, extrusion polymer expertise, and equipment solutions. With global operations and innovative PFAS-free materials, they deliver reliable, high-quality, and sustainable manufacturing services to help you prototype faster.
Explore Dynaflex Technologies’ polymer tubing on Chamfr, available in a range of materials.
Not finding what you need? Submit an RFQ directly to DynaFlex to share your custom compounding or specialty configurations.