Millstone Medical
Millstone Medical is the industry leader in sterile medical device manufacturing, offering product development engineering to accelerate time to market through pre-validated sterile packaging and integrated laboratory testing. With unmatched quality systems and regulatory expertise, Millstone enables faster, safer, and fully compliant product launches for the world’s leading OEMs.
Get Expert Guidance on Sterile Packaging & Testing

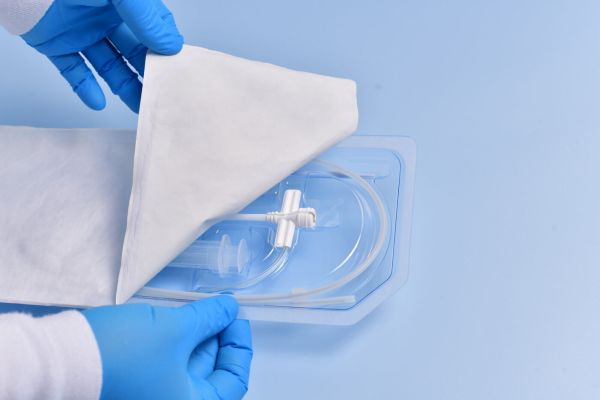
Pre-Validated Packaging & Fast Track Laboratory Testing
To support new product development time to market demands, Millstone offers the following solutions:
Pre-Validated Sterile Packaging
- Pre-validated PETG tray and pouch materials, including porous and non-porous options
- Five (5) year real-time aging validation already completed
- Compatible with multiple sterilization methods
- Double sterile-barrier options
Fast-Track New Product Lab Testing
- Bioburden testing & microbial identifications
- Sterility testing
- Endotoxin testing
- Cytotoxicity testing
- EO residuals
- Sterility & cleaning validations
One PO. From Suppliers to Finished Sterile Devices.
Speed To Market, Built Into Every Program.
- One program team under one PO, fewer handoffs across your launch path
- Supplier coordination and incoming inspection to keep components and timelines aligned
- Product-specific cleanroom assembly, cleaning, sterile packaging, testing, and distribution to support predictable release
We coordinate multiple suppliers, assemble, clean, package, test, and distribute your sterile devices.

Ready to Accelerate Your Time to Market?
Share your project needs with Millstone Medical’s experts.
