Blog
Expert insights and trends to accelerate medical device innovation and component sourcing.
Chamfr Resources
Learn. Explore. Connect.
New episodes, expert voices, delivered regularly.
As medical device companies scale, transferring manufacturing operations can be a pivotal move to ensure efficient, scalable, and compliant production that meets market demand. This sponsored article from Global Interconnect includes key considerations for a smooth manufacturing transfer.
With the addition of our newest supplier, Elringklinger, we’ve added nearly 50 new SKUs, providing R&D teams with a broader selection for their projects.
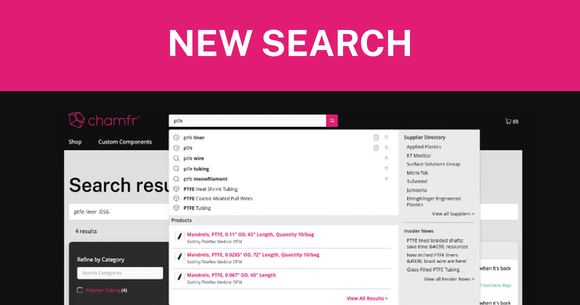
We know that every moment counts in your R&D process. That’s why we’ve upgraded Chamfr’s sitewide search to help you find the exact components and suppliers you need—faster and more efficiently than ever before.
We’re excited to introduce the latest addition to our equipment lineup – a hot box from our newest supplier, RTech!
Accelerate your medical device development with Bioana’s Design for Manufacturability (DFM) handbook. This comprehensive guide offers practical insights for various manufacturing processes to help you reduce iterations, cut development time and ensure a smoother path to success.
This week, new supplier KT Medical is bringing it with more braid wire and mandrel options, offering you greater choices on Chamfr!